
The two magnetic fields are usually chosen to be perpendicular to each other as this maximizes the NMR signal strength. After an RF pulse, precession usually occurs with the nuclei's intrinsic Larmor frequency and, in itself, does not involve transitions between spin states or energy levels. The detection of the NMR signal during or after the RF pulse, due to the voltage induced in a detection coil by precession of the nuclear spins around B 0.The oscillation frequency required for significant perturbation is dependent upon the static magnetic field ( B 0) and the nuclei of observation. 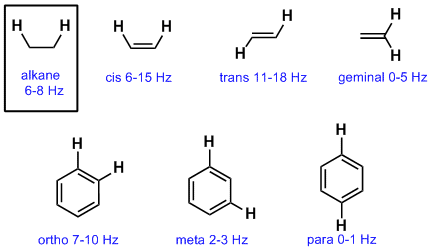
The perturbation of this alignment of the nuclear spins by a weak oscillating magnetic field, usually referred to as a radio frequency (RF) pulse.The alignment (polarization) of the magnetic nuclear spins in an applied, constant magnetic field B 0.The principle of NMR usually involves three sequential steps: Since the resolution of the imaging technique depends on the magnitude of the magnetic field gradient, many efforts are made to develop increased gradient field strength. It is this feature that is exploited in imaging techniques if a sample is placed in a non-uniform magnetic field then the resonance frequencies of the sample's nuclei depend on where in the field they are located. Nuclides with even numbers of both have a total spin of zero and are therefore NMR-inactive.Ī key feature of NMR is that the resonant frequency of a particular sample substance is usually directly proportional to the strength of the applied magnetic field. This occurs when an isotope has a nonzero nuclear spin, meaning an odd number of protons and/or neutrons (see Isotope). In order to interact with the magnetic field in the spectrometer, the nucleus must have an intrinsic nuclear magnetic moment and angular momentum. , can be studied by high-field NMR spectroscopy as well. , although isotopes of many other elements, such as 19 NMR is also routinely used in advanced medical imaging techniques, such as in magnetic resonance imaging (MRI). Nuclear magnetic resonance spectroscopy is widely used to determine the structure of organic molecules in solution and study molecular physics and crystals as well as non-crystalline materials. NMR results from specific magnetic properties of certain atomic nuclei. 20 tesla, the frequency is similar to VHF and UHF television broadcasts (60–1000 MHz). This process occurs near resonance, when the oscillation frequency matches the intrinsic frequency of the nuclei, which depends on the strength of the static magnetic field, the chemical environment, and the magnetic properties of the isotope involved in practical applications with static magnetic fields up to ca. Nuclear magnetic resonance ( NMR) is a physical phenomenon in which nuclei in a strong constant magnetic field are perturbed by a weak oscillating magnetic field (in the near field ) and respond by producing an electromagnetic signal with a frequency characteristic of the magnetic field at the nucleus. The signal at 3.Nuclear Magnetic Resonance (NMR) basic principles In this and in many spectra to follow, we show enlargements of individual signals so that the signal splitting patterns are recognizable. Rather than being a complication, however, this splitting behavior actually provides us with more information about our sample molecule.Ĭonsider the spectrum for 1,1,2-trichloroethane. In fact, the 1H-NMR spectra of most organic molecules contain proton signals that are 'split' into two or more sub-peaks. The 1H-NMR spectra that we have seen so far (of methyl acetate and para-xylene) are somewhat unusual in the sense that in both of these molecules, each set of protons generates a single NMR signal. Remember the n + 1 rule and the associated coupling patterns. Spin-spin coupling is often one of the more challenging topics for organic chemistry students to master. The split peaks ( multiplets) arise because the magnetic field experienced by the protons of one group is influenced by the spin arrangements of the protons in an adjacent group. This complication, which may be disturbing to a student who longs for the simple life, is in fact very useful to the organic chemist, and adds greatly to the power of NMR spectroscopy as a tool for the elucidation of chemical structures. True, we see absorptions in the regions we predicted, but these absorptions appear as a group of two peaks (a doublet) and a group of three peaks (a triplet). However, when we look at the spectrum it appears to be much more complex. \)įrom what we have learned about 1H NMR spectra so far, we might predict that the spectrum of 1,1,2-trichloroethane, CHCl 2CH 2Cl, would consist of two peaks-one, at about 2.5-4.0 δ, expected for CH 2-halogen compounds and one shifted downfield because of the presence of an additional electronegative chlorine atom on the second carbon.
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
